Personnel de l'université
Daniel MAUVOISIN
Coordonnées
- Tél
- 0228080010 (n° interne : 320010)
- Daniel.Mauvoisin@univ-nantes.fr
- Site internet
- https://orcid.org/0000-0003-0571-0741
Thèmes de recherche
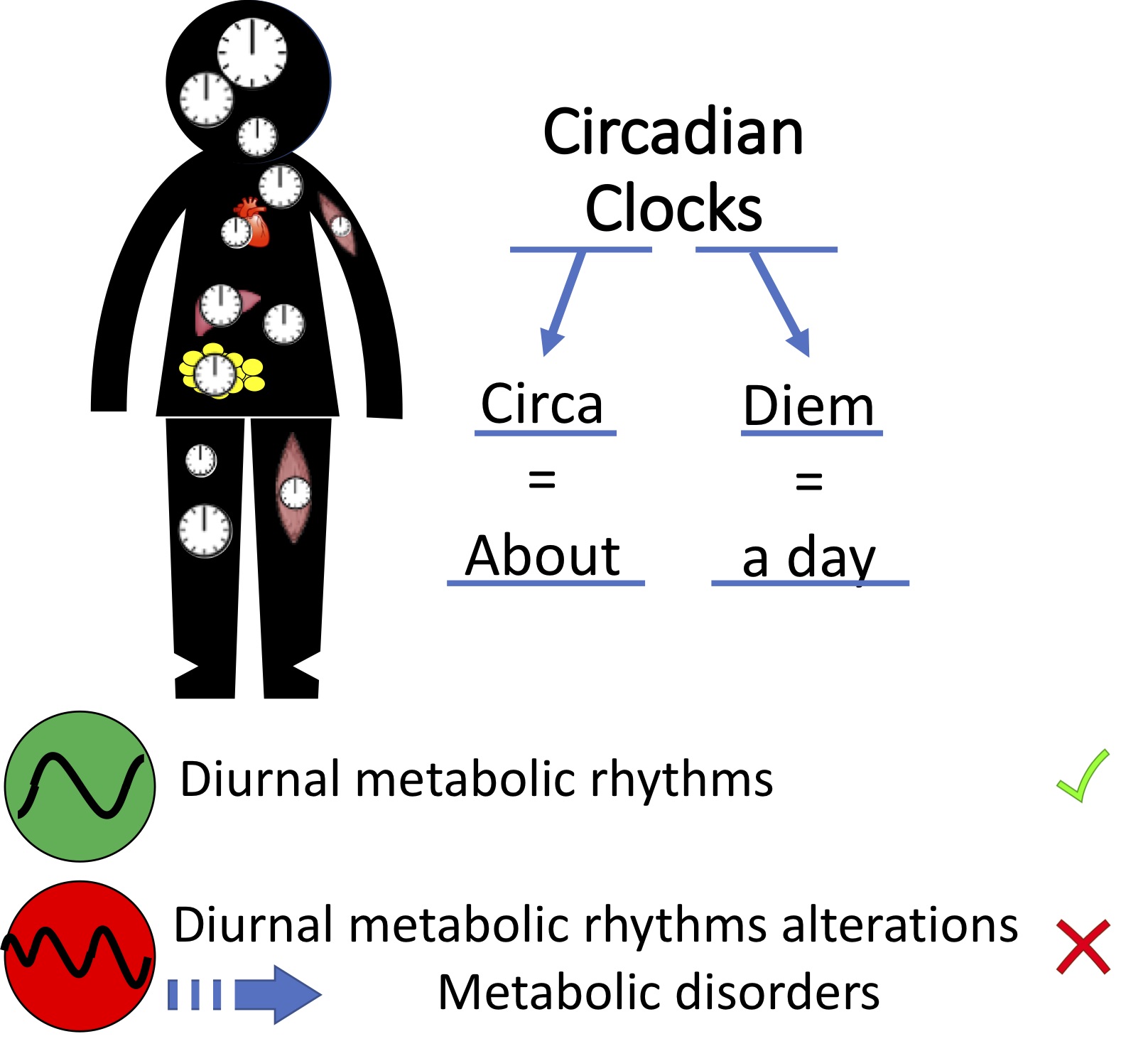
Modern lifestyles have contributed to a growing prevalence of chronic non-communicable diseases. In addition to diet quality and quantity, increasing evidence highlights another critical factor: when we eat. Irregular sleep patterns, disrupted daily routines, and erratic eating schedules act as circadian disruptors that increase the risk of overweight, metabolic dysfunction, and related diseases. As a result, the idea that “it’s not only what and how much we eat, but also when we eat” has gained significant attention. However, translating this concept into effective clinical strategies still requires a deeper understanding of the underlying biological mechanisms.
At the heart of this process lies the circadian clock, an internal time-keeping system that orchestrates daily physiological and metabolic rhythms. This hierarchical regulatory network plays a central role in maintaining metabolic balance, and its disruption has been linked to a wide range of conditions, including obesity and type 2 diabetes. The circadian clock regulates metabolism through multiple mechanisms, from gene transcription to post-translational modifications. At the cellular level, it also coordinates the dynamic organization of subcellular structures, such as daily rhythms in mitochondrial networks, which are essential for maintaining metabolic homeostasis.
Circadian regulation interacts closely with metabolic signals. Hormones, nutrients, redox sensors, and metabolites all act as outputs of the clock and, in turn, can feed back to influence it. Among these signals, feeding rhythms represent a powerful external cues (zeitgebers) capable of synchronizing metabolic processes with the daily cycle. Studies have shown that unrestricted consumption of high-fat diets disrupts natural feeding rhythms, whereas restricting food intake to the active phase can protect against metabolic dysfunction and liver pathologies, even in the context of diet-induced obesity.
A major challenge in the field of circadian metabolism is to identify the molecular mechanisms that connect feeding schedules with metabolic regulation across the day–night cycle. In particular, it remains unclear whether overnutrition—known to disrupt circadian rhythms—promotes metabolic disease by altering the rhythmic patterns of metabolic post-translational modifications and mitochondrial function.
Our research aims to uncover these molecular mechanisms, providing new insights into how circadian biology and nutrition timing interact. Ultimately, this knowledge will help translate circadian science into practical strategies to prevent and treat metabolic diseases and support healthier lifestyles.